Introduction
Mycobacterium tuberculosis (Mtb) remains one of the most lethal human pathogens, responsible for significant global mortality, with tuberculosis (TB) causing approximately 1.5 million deaths in 2020 alone. Although TB is most prevalent in low-income countries, its incidence is rising in Europe. Standard treatment for drug-susceptible TB involves prolonged multidrug regimens; however, the emergence of multidrug-resistant and extensively drug-resistant strains (MDR/XDR-TB) has complicated therapy, requiring longer, more toxic treatments with limited efficacy.
The persistence of TB is largely due to multiple resistance mechanisms, including antibiotic target modification, drug inactivation, efflux systems, and biofilm formation. Additionally, the unique structure of the mycobacterial cell envelope and the formation of granulomas hinder drug penetration and facilitate immune evasion. These challenges highlight the urgent need for novel therapeutic strategies.
β-lactam antibiotics, traditionally considered ineffective against Mtb, have recently regained attention. While earlier studies suggested intrinsic resistance, more recent clinical evidence demonstrates that combinations including carbapenems and β-lactamase inhibitors can significantly reduce bacterial burden. The limited efficacy of β-lactams is primarily due to poor permeability across the complex cell envelope and degradation by the β-lactamase enzyme BlaC.
Current research focuses on overcoming these barriers through combination therapies and novel approaches. One promising strategy involves targeting the extracellular glycan-rich capsule, which plays a critical role in Mtb survival and pathogenicity. The BAN-BOOT project aims to develop dual-action molecules capable of inhibiting β-lactamase activity while disrupting the capsule structure, thereby enhancing antibiotic penetration. Further optimization through lipid-based prodrug design is expected to improve drug delivery to granulomas and combat resistance more effectively.
Background of the project
For several years, our group has been working on the synthesis of boron-containing compounds as inhibitors of Serine-Beta-Lactamases (SBLs). Some of these compounds were tested against the BlaC enzyme, and a few showed good activity against this enzyme, in particular the compound called EC19 showed an inhibition constant of 0.65 micromolar.
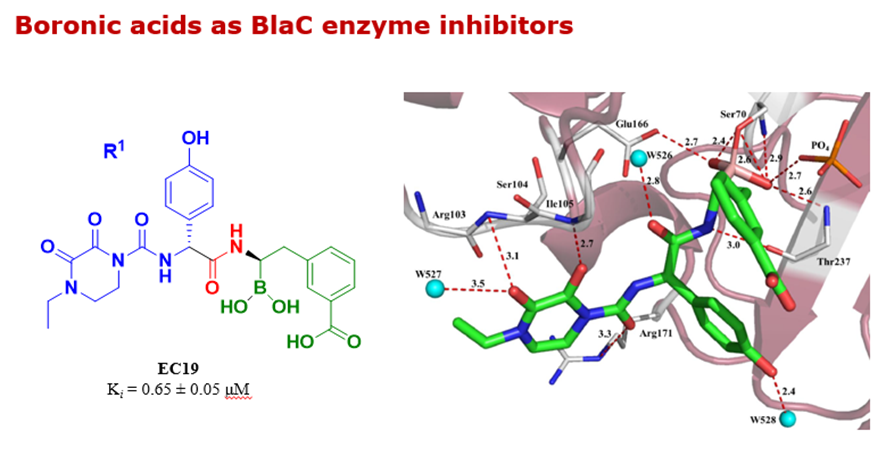
According to the X ray structure, the boron atom forms a covalent bond with the catalytic serine in analogy to other boron-based inhibitors. In addition, one of the boronate oxygen interacts with a serine and a threonine. The other boronate oxygen replaces the hydrolytic water and makes a hydrogen bonding interaction with a key glutamic residue, involved in the hydrolytic mechanism. The substituent in blue makes several interactions with active site residues, such as a serine, and arginine.
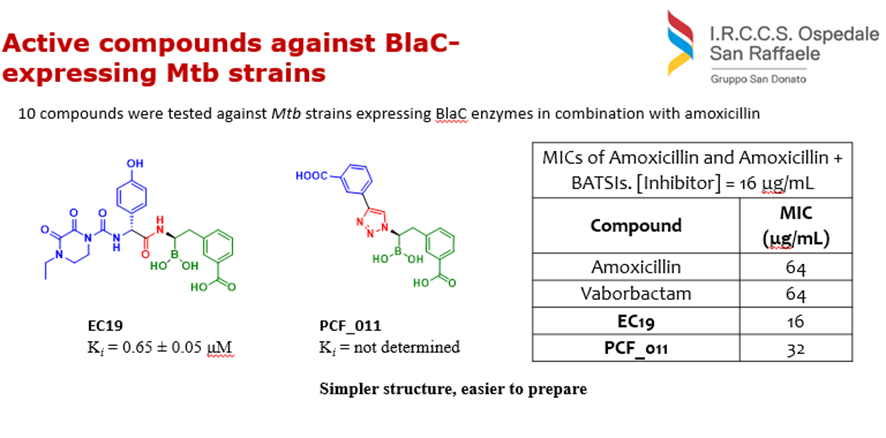
Considering this precedent, last year our group has started a collaboration with Prof Cirillo’s Lab at San Raffaele Hospital. From a library of 300 BATSIs previously synthesized, we chose 10 compounds that were tested against Mtb strains expressing BlaC enzymes in combination with amoxicillin. Two of these compounds proved to slightly increase the amoxicillin susceptibility against BlaC-producing Mtb reference strains. One of them, the known BlaC inhibitor EC19, and PCF_011, a simpler compound and synthetically easier, showed good results against Mtb.
One of the problems to achieve Mtb is to cross the Mtb cell envelope which is very complex and rich in carbohydrates and lipids that prevent intracellular access to many molecules.
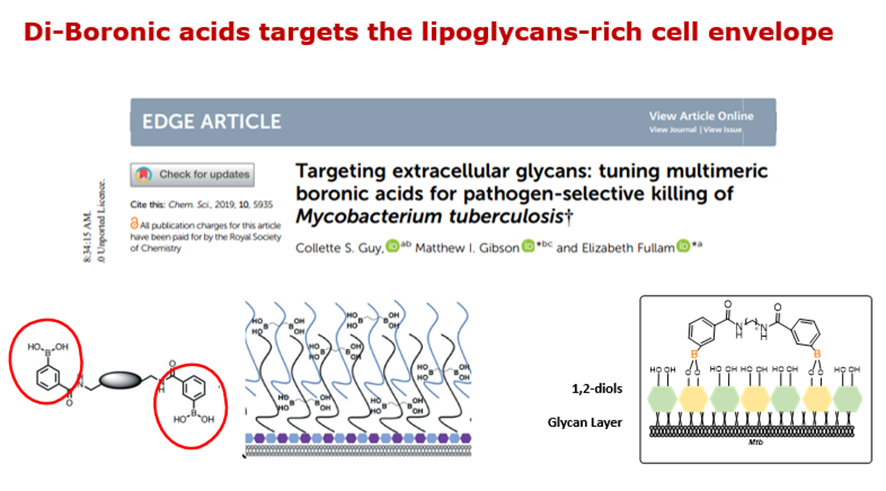
And in this sense, in Fullaman’s work it was described some di-boronic acids compounds able to form covalent cis-1,2-diols of the glycan layer polysaccharides present in the cell envelope. Thus, these agents are able to weaken and disrupt this glycan layer in the external Mtb cell envelope, which is essential for them, so they showed potent antibacterial activity.
BAN-BOOT aims
The general aim of BAN-BOOT is to develop boron-based compounds for an innovative anti-tubercular strategy.
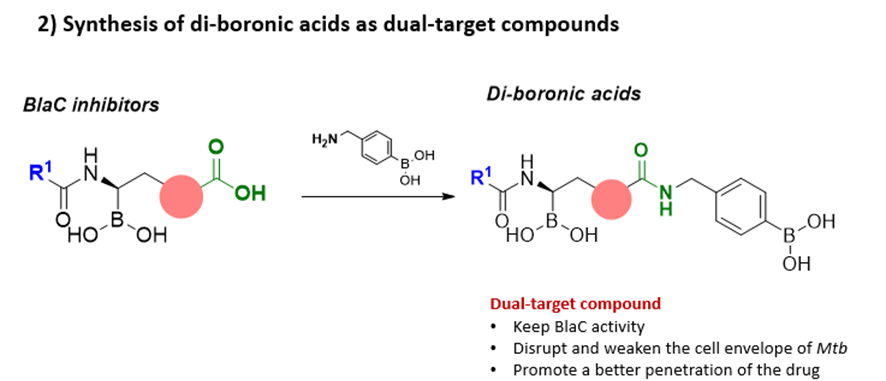
Specifically, the first aim is to synthesize new BlaC inhibitors more potent than the ones already tested, bearing the boronic acid, an aromatic ring, or a triazole group that is synthetically easy to prepare thought a click reaction, between an azide and an alkyne, a carboxylic acid, and bearing different antibiotic side chains.
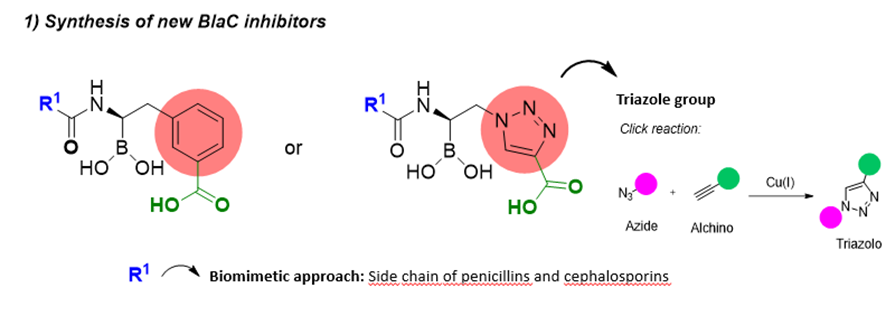
Once identified the best BlaC inhibitors, the carboxylic group is modified though an amide synthesis with a benzylamine containing a phenyl boronic acid to give a di-boronic acid compound. This strategy create compounds which directly disrupt and weaken the cell envelope of Mtb therefore promoting a better penetration of the drug, while keeping the activity against BlaC enzymes.
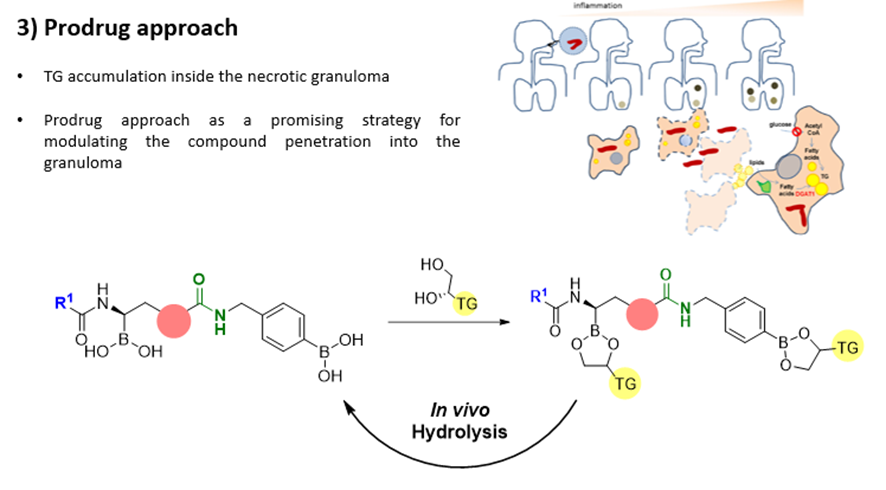
During the disease development, it is formed a granuloma that acts as a barrier and prevent the drug diffusion into the infected site. One interesting characteristic of Mtb is that, during the infection, it can change its metabolism, reducing the up-take of glucose and increasing the up-take of lipids. By preparing prodrugs, the boronic acid can be derivatized with a diol bearing the triglycerides to give the lipid-prodrug that could be hydrolyzed in vivo and release the active compound. This approach should improve the compound penetration into the granuloma and thus to improve the in vivo activity.
Overall, BAN-BOOT aims to obtain new molecules that could serve as BLA adjuvant with a double action: by inhibiting BlaC, the BL expressed in Mtb, and by disrupting the external “capsule” comprised of a glycan layer, therefore promoting a better penetration of the drug. Once identified a highly active compound against Mtb, a lipid-based prodrug derivatization will be attempted to favor the diffusion into the mycobacteria-harboring centrally granuloma. By using a multi-strategy approach, the drug resistance development will be tackled in an efficient manner.
